 There are exceptions however, such as water's density increasing between 0☌ and 4☌.īelow is a table of units in which density is commonly expressed, as well as the densities of some common materials. Increases in temperature tend to decrease density since the volume will generally increase. An increase in pressure decreases volume, and always increases density. However, when regarding gases, density is largely affected by temperature and pressure. In the case of solids and liquids, the change in density is typically low. Note that density is also affected by pressure and temperature. It is useful to carefully write out whatever values are being worked with, including units, and perform dimensional analysis to ensure that the final result has units of mass There are many different ways to express density, and not using or converting into the proper units will result in an incorrect value. However, it is important to pay special attention to the units used for density calculations. If you are curious about the density of your dish soap, try the simple experiment mentioned above to determine it.The calculation of density is quite straightforward. Knowing the density of your dish soap can help you determine the amount needed to effectively clean your dishes while avoiding waste. In conclusion, the density of dish soap is an important factor to consider when using this common household item. Use the formula D=M/V to find the density of the bar of soap. Water - Density, Specific Weight and Thermal Expansion Coefficients Definitions, online calculator and figures and tables with water properties like density, specific weight and thermal expansion coefficient of liquid water at temperatures ranging 0 to 360☌ (32 to 680☏). Then use the double arm balance to find the mass of the bar of soap. VIDEO ANSWER: The density of water is equal to the mass divided by the volume, so the density will be 1000 kilogram per meter cube. Density of sulfuric acid at various temperatures and concentrations. The density of distilled water is the same. 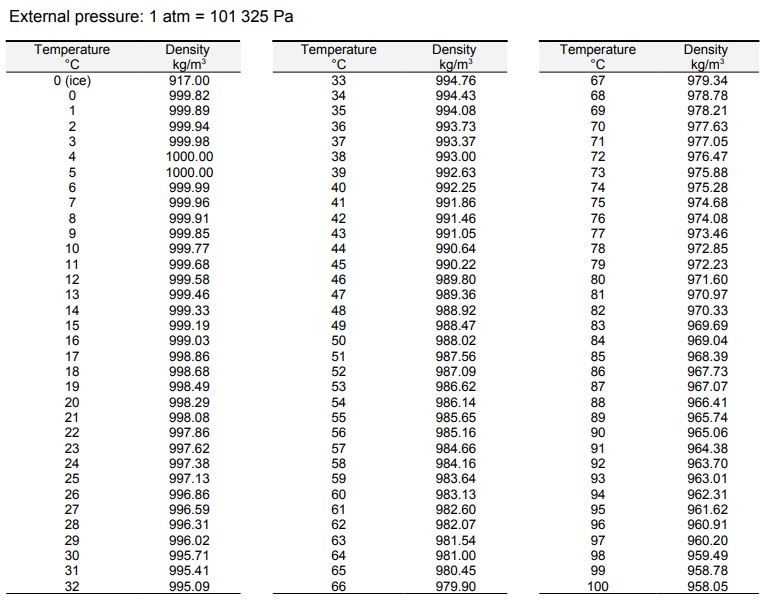 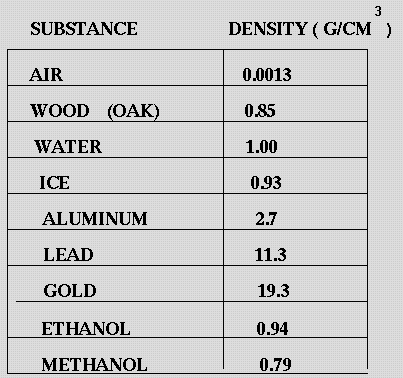
At room temperature, water remains in a liquid state. The density of water at 25 degrees Celsius is 997 kg/m3. At room temperature ( 200C), its value is 998.2 kg/m3. The density of water is defined similarly to other substances. Multiply the width x length x height to find the volume of the bar of soap. The Density of Water At Room Temperature. Use your ruler to measure the length, width, and height of the bar of soap. If you take a bar of soap made of just soap ingredients, it is denser than the water in your tub and therefore sinks. 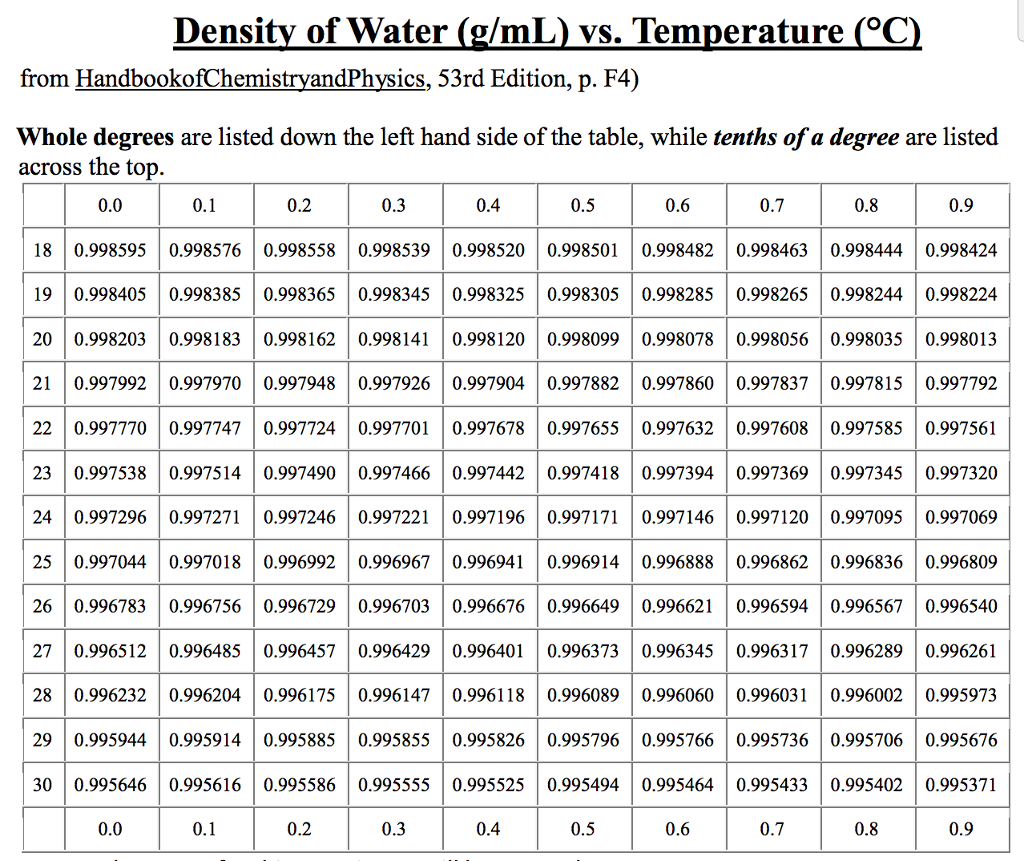
One of the characteristics of buoyancy is surface area density. What Is The Density Of Dish Soap In Kg M3?ĭishwashing Liquid 1 120 kg / m3. Let’s find out the densities by visiting Denseme FAQ What Is The Density Of Dish Soap In Ml? The density and value of gold depend on its 'karat' value, which is a measure of how many parts in 24 (by mass) are pure gold. Calculate the density of the soap using the formula Density = (mass of soap and water – mass of water)/volume of water.Weigh the cylinder with the water and soap mixture and record the mass.Add a small amount of dish soap to the water and stir gently. 
Weigh the cylinder with the water and record the mass.Fill the graduated cylinder with a known volume of water, for example, 100 mL.All you need is a graduated cylinder, some dish soap, and a scale. If you are curious about the density of your dish soap, you can perform a simple experiment at home. On the other hand, using too little soap may not effectively clean your dishes. Using too much soap can lead to waste and potential damage to your dishes. Understanding the density of the soap can help you determine the amount of soap needed to get the job done. Why Does Density Matter?ĭensity is an important factor to consider when using dish soap. For example, some concentrated dish soaps have a higher density than regular ones, meaning you need less of the product to get the same cleaning power. However, this can vary from brand to brand and even between different types of dish soap. On average, dish soap has a density of around 1.06 grams per milliliter (g/mL). The density of dish soap varies depending on its brand and type. The formula for density is Density = Mass/Volume. In simple terms, it is how heavy something is for its size. Density is the measurement of how much mass is contained in a given volume. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
